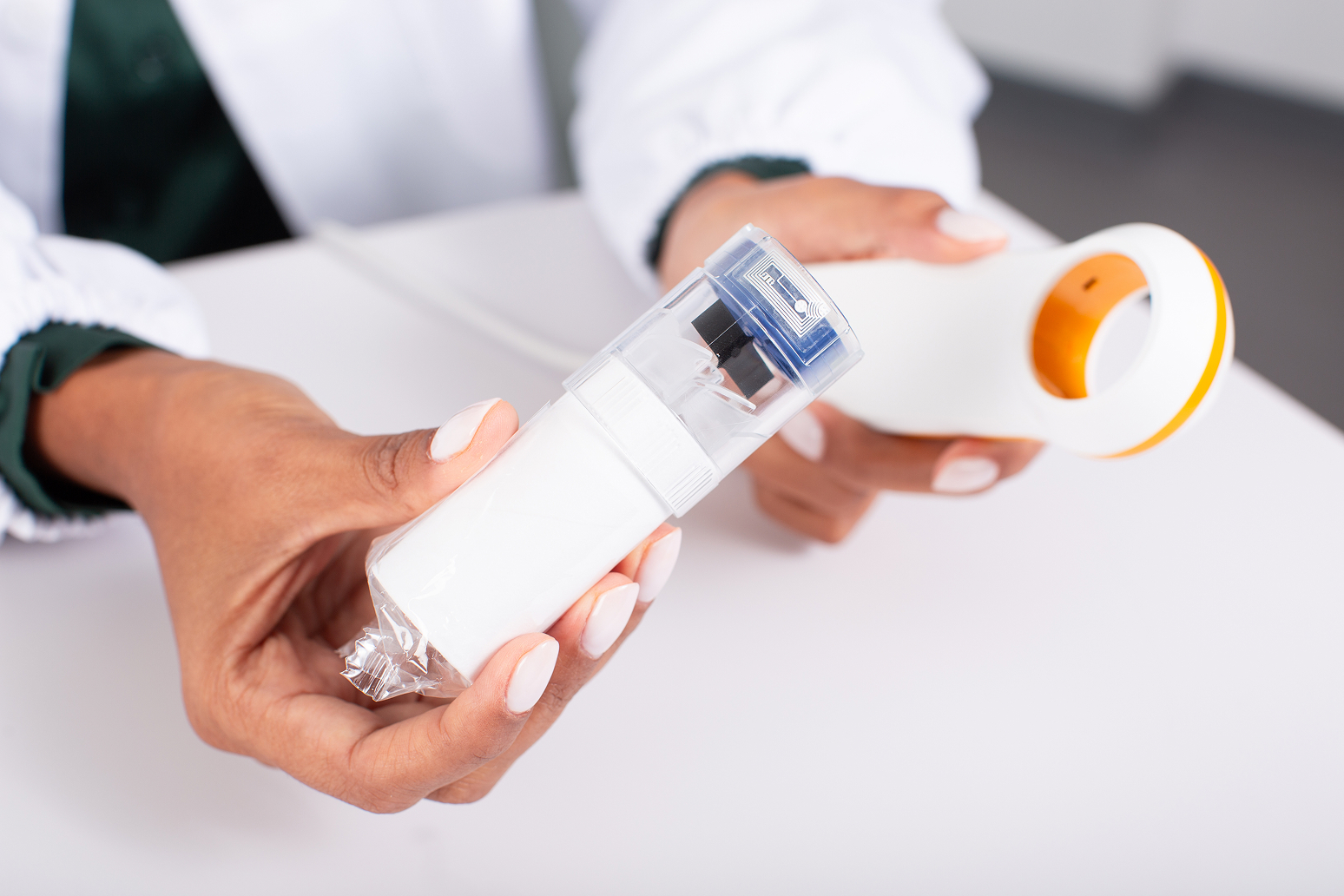
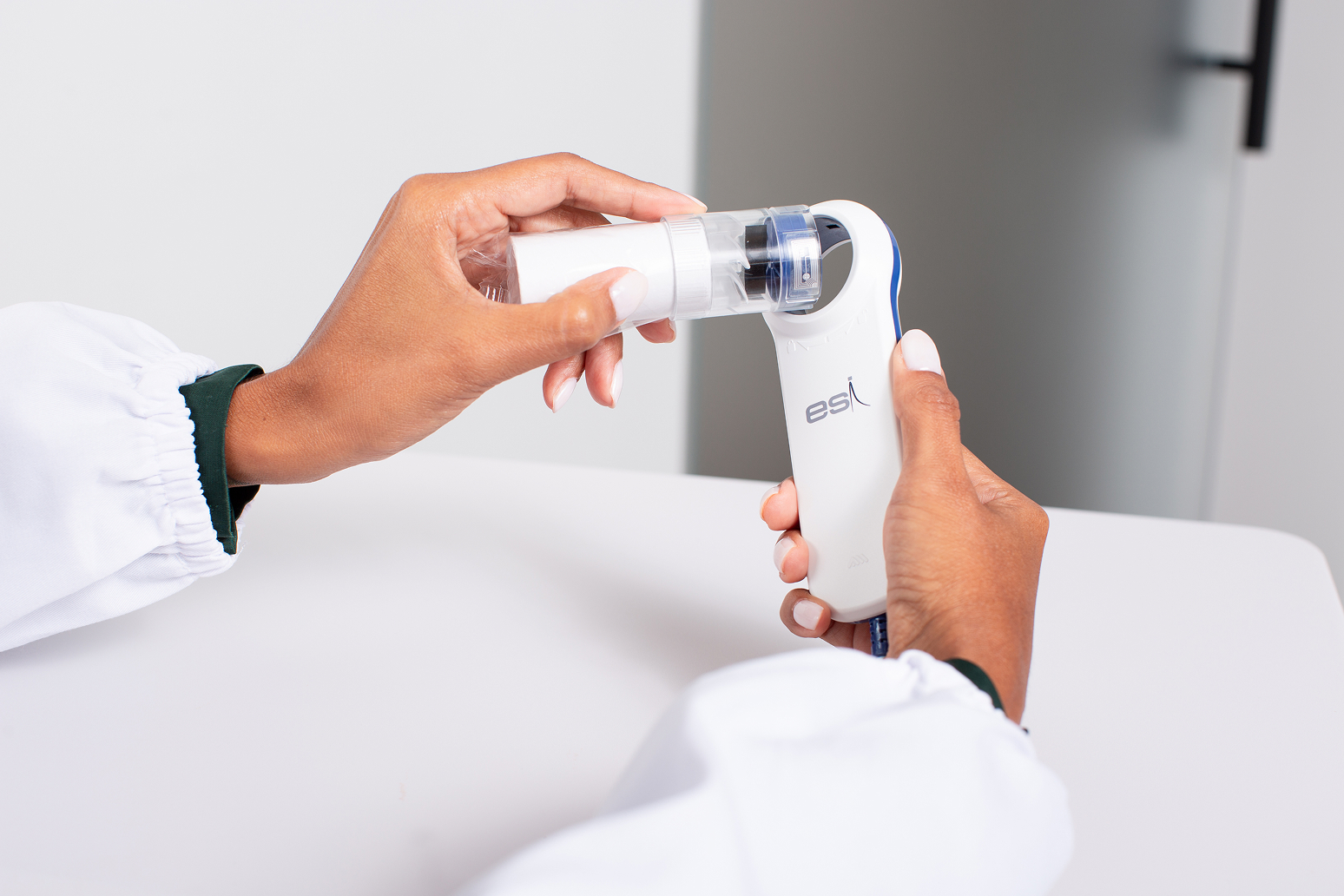
Flowmir Plus, Disposable Turbine
Disposable turbine with integrated RFID technology, individually packaged
-
-
Ultra-high sensitivity to low flows (20 mL/s)NEW
-
High repeatability and accuracy
-
ISO 26782 and ISO 23745 compliance
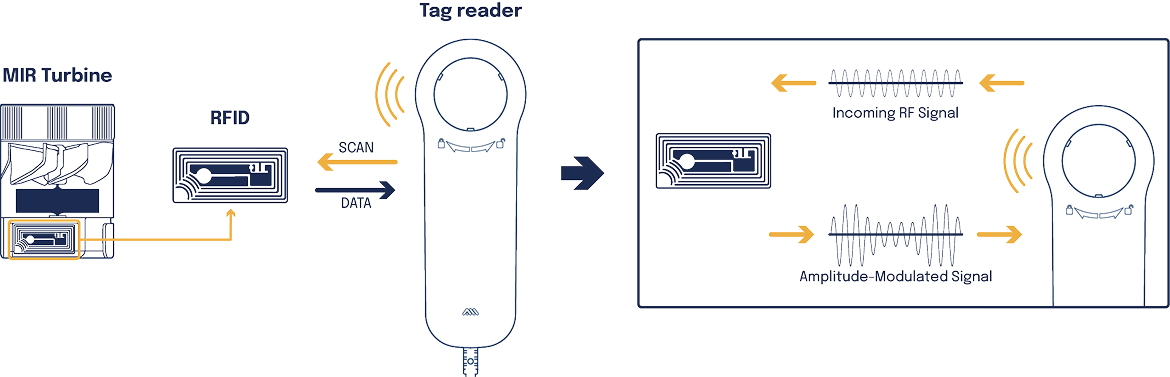
Flowmir Plus is the first disposable turbine to establish direct communication with MIR’s professional devices. The built-in reading and writing system allows Flowmir Plus to transmit crucial measurement data, such as calibration and automatic turbine type recognition, via RFID.
This technology simplifies the device’s usage and management process, minimizing the need for manual interventions and reducing the risk of errors. Ideal for maximum hygiene, disposable turbines are the perfect solution for those seeking simplicity and convenience. Factory-calibrated and ready to use, they eliminate the need for further calibration, cleaning, or maintenance.
Flowmir Plus turbines can be used either with disposable paper mouthpieces or with the Neumofilt Ergo antibacterial filter, available separately.
Clinical Evidence
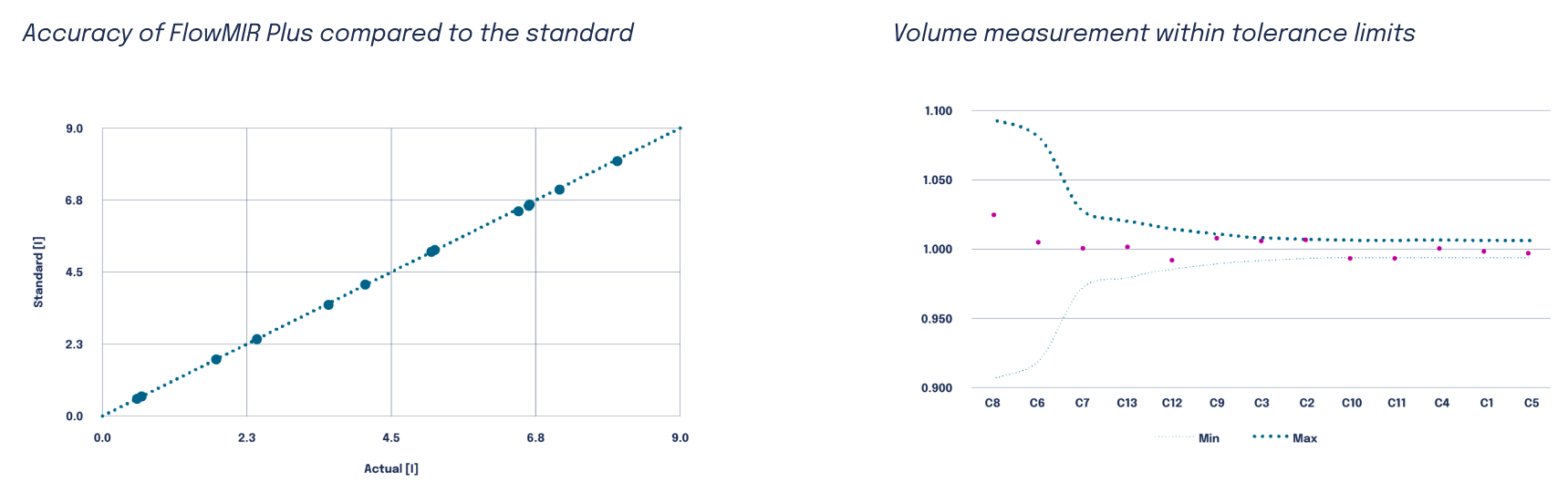
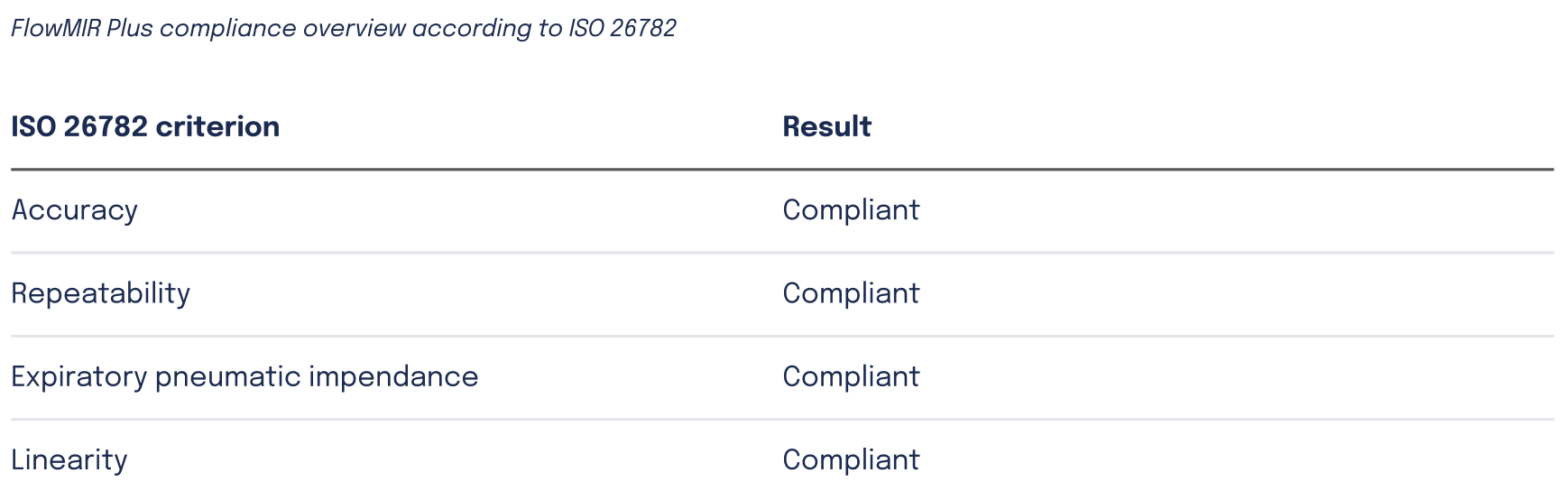
Flowmir Plus has been evaluated in a dedicated compliance assessment according to ISO 26782, the international standard that defines performance requirements for spirometers intended for the measurement of time-forced expired volumes in humans.
The study was conducted using the Flowmir Plus disposable turbine, the Spirobank II Plus professional spirometer, and a pulmonary waveform generator compliant with ATS/ERS guidelines. The testing protocol included repeated measurements across the 13 Defined Test Profiles C1–C13, which are designed to reproduce different forced expiratory patterns observed in clinical practice.
The analysis focused on three key spirometric parameters: FEV₁, FEV₆, and FVC. Flowmir Plus performance was assessed according to four technical criteria: accuracy, repeatability, expiratory pneumatic impedance, and linearity. The results confirmed that Flowmir Plus measurements complied with the limits established by ISO 26782 for all evaluated criteria.
Compatibility
Flowmir Plus can only be used with MIR Plus professional devices
Available in dispenser boxes of 20 (code: 91300120) or 60 units (code: 91300160)
All the features
-
High sensitivity to low flowsNEW
Plus Turbines ensure accurate measurements, even with extremely reduced lung capacity (20 mL/s)
-
Calibration stored on RFIDNEW
The innovative integrated RFID system records calibration data, ensuring measurement accuracy and reducing maintenance tests between patients
-
Automatic turbine type recognitionNEW
The integrated RFID system allows for automatic recognition of the turbine
-
No influence from environmental variations
Research studies demonstrate the turbine sensor’s reliability in various environmental conditions. The turbine is unaffected by changes in pressure, humidity, temperature, and air viscosity
-
Automated testing and assemblyNEW
The fully automated testing and assembly processes provide maximum safety, precision, and reliability for each unit
-
Individually packed
The individual packaging of the turbine is essential for preserving hygiene, reducing the risk of cross-contamination, and enhancing overall quality
-
High repeatability and accuracy
Plus Turbines deliver consistent results with no variations in measured data
-
ISO 26782 and ISO 23747 compliance
Become a Dealer
Interested in Becoming a Dealer? Learn more on the requirements and how to become an official MIR dealer.
Infection control

MIR turbines overview

ATS / ERS |

Flowmir Plus, Disposable Turbine
|

Reusable Turbine Plus
|

Reusable Turbine
|

FlowMIR®, Disposable Turbine
|
|---|---|---|---|---|
|
ATS / ERS |

Flowmir Plus, Disposable Turbine
|

Reusable Turbine Plus
|

Reusable Turbine
|

FlowMIR®, Disposable Turbine
|
|
Compatible Spirometers |
||||
|
Mouthpiece |
To be purchased separately
|
To be purchased separately
|
To be purchased separately
|
Included
|

RFID |
Automatic turbine type recognition, Calibration stored on RFID, Turbine traceability
|
Automatic turbine type recognition, Calibration stored on RFID, Turbine traceability
|
No
|
No
|
|
Antiviral Filter |
Not required
|
To be purchased separately
|
To be purchased separately
|
Not required
|
|
Turbine Disinfection |
Not Required
|
Required
|
Required
|
Not required
|
|
Turbine Calibration |
Not required but available at any time
|
Not required for first use but available at any time
|
Not required but available via software at any time
|
Not required but available via software at any time
|
|
Packaging |
Individually sealed in carton dispenser, 60 pcs or 20 pcs
|
Individually sealed in carton box
|
Carton box
|
Individually sealed in carton dispenser (60pcs or 10pcs)
|
|
Metal mesh |
None
|
None
|
Included
|
Not required
|
|
Turbine Lifetime |
Single use, no expiration date
|
5 years
|
5 years
|
Single use (no expiration date)
|
|
Maintenance Cost |
None
|
High
|
High
|
None
|
Looking for our Company and Product Certificates and Compliance Documents?
Ask for information or support
If you need more information about the product or you need some help, fill the form.