Spirolab Plus ESI
Detection of Emphysema by Spirometry
-
-
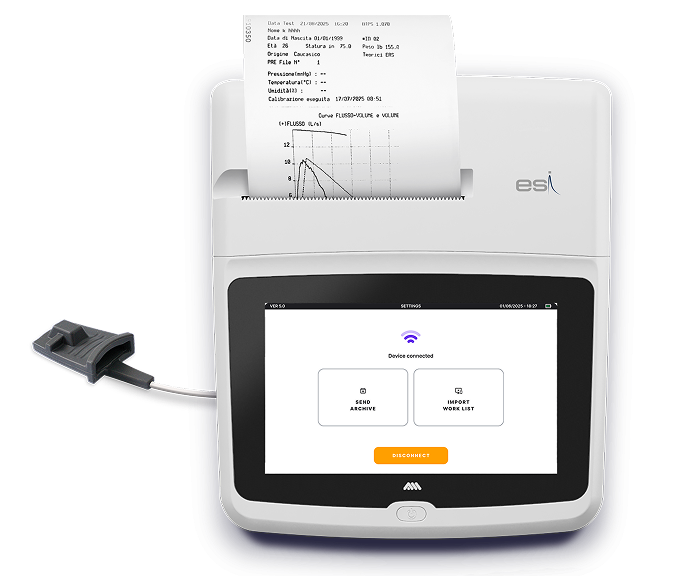
Wi-FiNEW
-
Work ListNEW
-
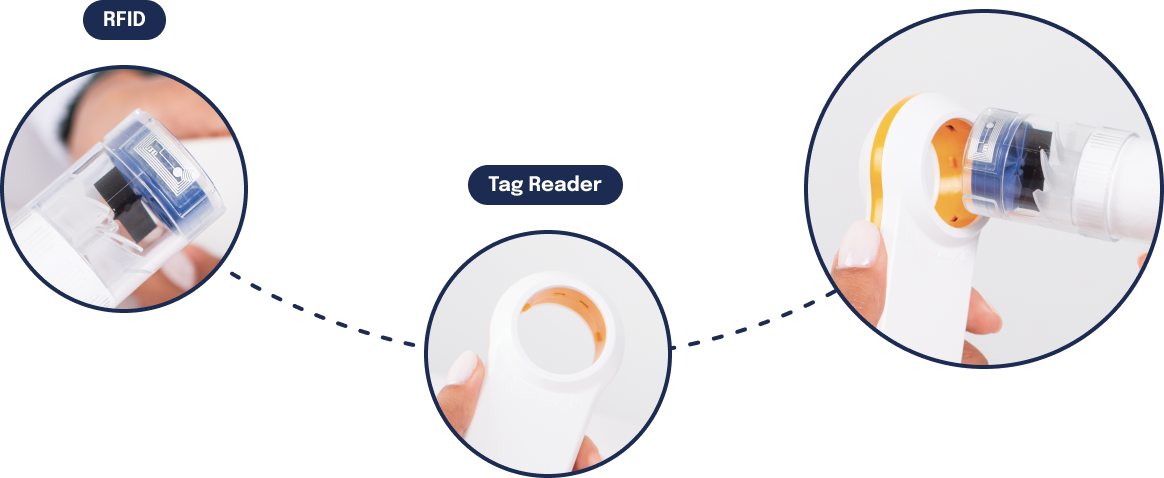
RFID TurbineNEW
-
EMR Integration
-
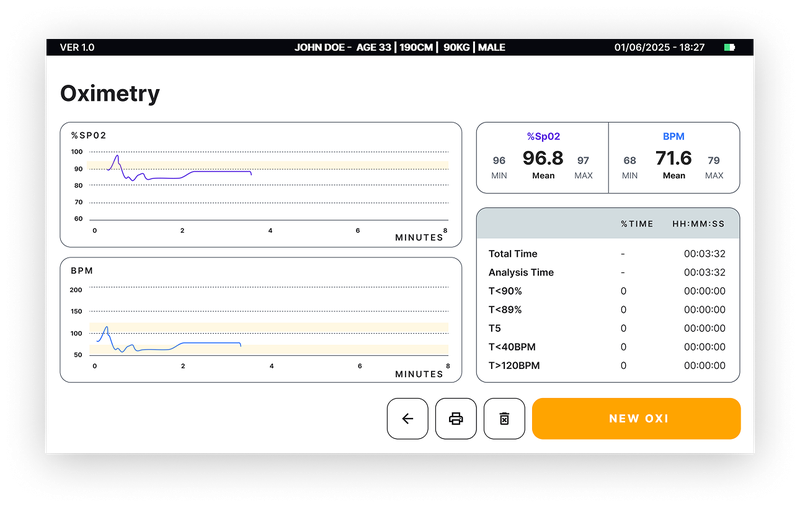
OximetryOptional
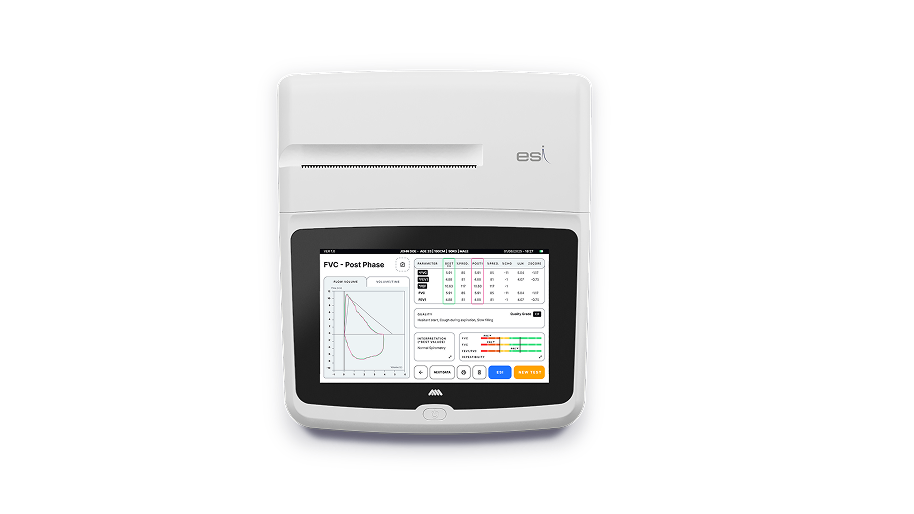
Spirolab Plus ESI is the first desktop spirometer to detect emphysema by spirometry. The embedded algorithm computes ESI value along with the other standard spirometry parameters. The computation of ESI – Emphysema Severity Index, is based only on the morphology of the Flow/Volume Curve of each subject, independently of its own age, sex, ethnic and anthropometric characteristics.
ESI value allows to define in each patient, by a standard spirometry, whether airflow obstruction is due to inflammatory narrowing of the conductive airways (chronic bronchitis, bronchiolitis, small airway disease) or to the destruction of lung parenchyma caused by Emphysema. Identifying the cause of airway obstruction is crucial in the diagnosis and treatment of Chronic Obstructive Pulmonary Disease (COPD).
Patients with the same level of obstruction (FEV1) may reflect very different underlying conditions.
ESI makes this difference visible, supporting targeted therapeutic strategies.
Spirolab Plus ESI is compliant with the latest international standards and recommendations for spirometry and oximetry, including ATS/ERS 2019-2021 guidelines, ISO 26782:2009, ISO 23747:2015, ISO 80601-2-61, and other applicable international standards.